With increasing bacterial resistance, companies are pulling out of antibiotic research and fewer new antibiotics are approved

Antibiotic overprescribing in both hospital and community settings has significant consequences – often prescribed unnecessarily for viral upper respiratory tract infections
Most overprescribing for respiratory infections is in Primary Care – in particular for patients presenting with an acute cough
- Resistance – resistant organisms/superbugs
- Changes in normal microbial flora – mild – thrush, severe and sometimes life-threatening – Clostridium difficile
- Side effects of medication
- In current times of austerity cost also plays a part
The consequences of indiscriminate antibiotic prescription
| Patient-level | Community-level |
| • Adverse events | • Increased antibiotic resistance |
| • Unnecessary cost | — Infections becoming more difficult |
| • Drug-drug interactions | — Epidemics harder to control |
| • Increased risk of infection with an antibiotic-resistant strain | • Increased healthcare cost |
| • Fewer effective therapeutic options |
Principles of appropriate antibiotic prescribing TREAT: Bacterial infection only OPTIMISE: Diagnosis/severity assessment MAXIMISE: Bacterial eradication only (or reduction) RECOGNISE: (Local) resistance prevalence UTILISE: PK/PD — effective choice agent and dose INTEGRATE: Local resistance, effcacy, cost & effectiveness Appropriate prescribing conforms to these criteria
Treat Bacterial Infections only
Acute respiratory tract infections – When are antibiotics indicated?
Inappropriate prescribing of antibiotics for acute RTIs contributes to the increasing prevalence of antibiotic resistance.
- More than half of all antibiotics prescribed for acute RTIs are unnecessary because these infections are most likely viral and therefore not treatable with antibiotics
- Clinicians in ambulatory-care settings prescribe antibiotics for patients with a viral acute RTI in 40% to 50% of cases
- Studies have demonstrated that clinician and patient education is a vital component of reducing inappropriate antibiotic use
Clinical Practice Guideline (Update): Adult Sinusitis
- Emphasis on patient education and counseling with new explanatory tables
- Extension of watchful waiting (without antibiotic therapy) as an initial management strategy to all patients with uncomplicated acute bacterial rhinosinusitis (ABRS) regardless of severity, not just patients with “mild illness” (prior guideline)
Colonization vs. infection: common clinical scenario
- Sputum/respiratory specimen showing organisms in a patient with suspected pneumonia.
- Assessing sputum quality uses a grading system based on neutrophils, epithelial cells, and mucus
- This helps to interpret results of sputum culture (distinguish commensals from saliva vs. pathogens from sputum)
Bartlett’s grading system for evaluating the quality of sputum samples.
| No. of neutrophils/low-power field | Grade |
| <10 | 0 |
| 10-25 | +1 |
| >25 | +2 |
| Presence of mucus threads | +1 |
| No. of epithelial cells/low-power field | Number |
| <10 | 0 |
| 10-25 | -1 |
| >25 | -2 |
Average the number of epithelial cells and neutrophils in about 20 or 30 separate low-power fields and then calculate the total. A final score of 0 or less indicates a lack of active inflammation or contamination with saliva.
Some common clinical scenarios which require this differentiation are : patients with burns, chronic wounds like osteomyelitis with/without a draining sinus and diabetic foot ulcers, cultures from postoperative drains, positive urine cultures in an asymptomatic patient and in a catheterised patient sputum/respiratory specimen showing organisms in a patient with suspected pneumonia, blood cultures in a patient with indwelling central venous catheter, arterial line, hemodialysis catheter.
The points which help distinguish colonization versus invasion are related to the findings at the site, the characteristics of the patient, the organism and on follow up.
OPTIMISE – ACCURATE DIAGNOSIS AND SEVERITY ASSESSMENT
Accurate clinical diagnosis is key to limit antibiotic prescribing
- Guidelines should offer practical criteria to identify those bacterial infections requiring antibiotic therapy
- Most guidelines recommend antibiotics to be reserved for moderate to severe infections
- A study from the United States (led by a family physician) shows that antibiotics are prescribed for 65% of episodes of URTI, 78% of acute bronchitis, 65% of acute pharyngitis, and 81% of acute sinusitis
• Determining the Likelihood of a Bacterial Infection More than 90% of otherwise healthy patients presenting to their outpatient providers with an acute cough have a syndrome caused by a virus • Appropriate Management Strategies A randomized, placebo.controlled trial (not included in the Cochrane review) comparing ibuprofen, amoxicillin-clavulanlc acid. and placebo showed no significant differences in the number of days to cough resolution Although macrolides azithromyCin are frequently prescribed for patients With a cough. one study showed that patients with acute bronchitis treated with a macrolide had significantly more adverse events than those receiving placebo. Clinicians should not perform testing or initiate antibiotic therapy in patients with bronchitis unless pneumonia is suspected
The Centor criteria are a set of criteria which may be used to identify the likelihood of a bacterial infection in adult patients complaining of a sore throat.
The Centor criteria are a set of criteria which may be used to identify the likelihood of a bacterial infection in adult patients complaining of a sore throat.
| Criteria | Points |
| Absence of Cough | 1 |
| Swollen and Tender Anterior Cervical Nodes | 1 |
| Temp of > 100.4• F (38•C) | 1 |
| Tonsillar Exudates or Swelling | 1 |
| Age | |
| 3-14 years | 1 |
| 15-44 years | 0 |
| 45+ | -1 |
Antibiotics should not be used in patients with a less severe presentation of sore throat, e.g. 0–2 Centor criteria to relieve symptoms
MAXIMISE
Therapy should reduce maximally or eradicate the bacterial load
Inappropriate therapy fails to eradicate the causative pathogens but may provide an environment that selects for resistant bacterial strains. Resistant bacteria may then spread, increasing the prevalence of resistance, the risk of resistance-related treatment failure and treatment costs. Without appropriate antibacterial therapy, this cycle of continued infection, resistance emergence, and treatment failure cannot be broken.
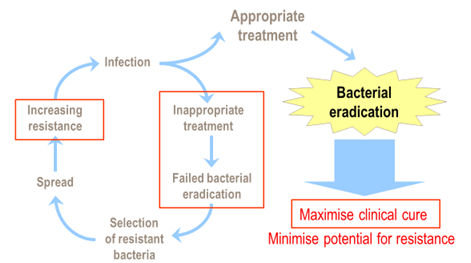
Appropriate antibacterial therapy results in bacterial eradication, thus maximizing clinical cure and minimizing the potential for the development and spread of resistance.
RECOGNISE- ANTIBIOTIC CHOICES MUST
REFLECT LOCAL RESISTANCE PREVALENCE
Take account of local susceptibility data and their resistance patterns when
selecting an antibiotic
For both hospitals and community
- Systematic monitoring and surveillance of antibiotic susceptibility of bacterial pathogens
- This information would enable authorities to design and implement early interventions that halt the spread of such strains
UTILISE- PK/PD – EFFECTIVE CHOICE OF AGENT AND DOSE
Apply pharmacodynamic parameters in choice of effective agents and dosage
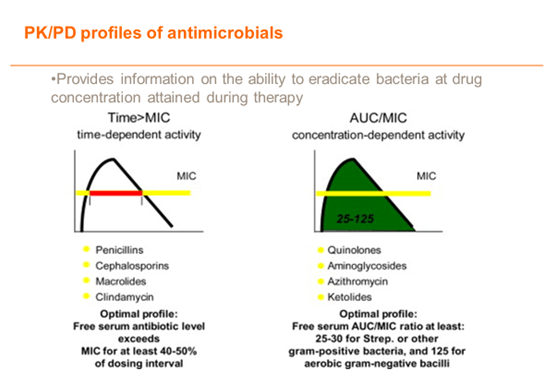
DURATION OF THERAPY
Defining the optimal duration of antibiotic
treatment is often a dilemma
- Longer than a necessary duration of antibiotics can result in:
- greater risk of toxicity, a higher rate of adverse events
- increased risk of resistance
- increased days of hospitalization, morbidity or mortality
- reduction in patient compliance – compliance is better with a shorter course
Meta-analyses of Acute Otitis Media (AOM), Acute Bacterial sinusitis (ABS), acute exacerbation of chronic bronchitis (AECB), Community-acquired pneumonia (CAP)
- AOM: 5 vs. 8–10 day therapy: No significant difference in clinical outcomes
- ABS: 3–7 vs. 6–10d NS (AE less in former)
- COPD: 5 vs. 7–10d NS (AE less in former)
- CAP: Adults: 3-7 vs. 7-10d: NS
- The above meta-analyses indicate that a short-duration treatment seems to be as effective as a longer course of antibiotic treatment in such infections as acute otitis media, acute bacterial sinusitis, CAP and infectious exacerbations of chronic bronchitis
GUIDELINES – Implementation in clinical practice
Reasons why antibiotic prescribing guidelines are not followed



Embrace antibiotic stewardship
- Improve antibiotic use in private practice as well as in hospitals—regardless of size—through stewardship interventions and programs, which will:
- improve individual patient outcomesreduce the overall burden of antibiotic resistancesave healthcare cost
- Refer to standard treatment guidelines
- Practice rational use of antibiotics

Sir, Excellent article
Sir, Can I share your article on my blog