Gene therapy
Gene therapy is an experimental treatment that involves introducing genetic material into a person’s cells to fight or prevent disease.
Approaches:
- Replacing a mutated gene that causes disease with a healthy copy of the gene.
- Inactivating, or “knocking out,” a mutated gene that is functioning improperly.
- Introducing a new gene into the body to help fight a disease
Types of Gene Therapy
- Germ line gene therapy
- Germ cells (sperm or egg) are modified by the introduction of functional genes, which are integrated into their genome.
- Inheritable changes
- Ethical issues – Gene has failed to be introduced or fails to be activated, the resulting child would likely be no worse off than he or she would have been without the attempted gene transfer
- Somatic gene therapy
- Genes with therapeutic potential are transferred into the somatic cells of a patient.
- Not inherited – Any alteration in genetics of particular persons will be restricted to the individual patient only and will not be inherited by the patient’s offspring or any later generation
- Approach for gene therapy studies
History – 1990
- First successful gene therapy study on Ashanti De Silva
- Adenosine deaminase (ADA) deficiency – SCID – Severe combined immunodeficiency syndrome
- WBCs extracted, genes producing ADA implanted into WBCs and then transferred the cells back to her body.
- The WBCs strengthened the girl’s immune system made it possible for her to survive.

Direct -> InVivo
Indirect/Cell based à ExVivo
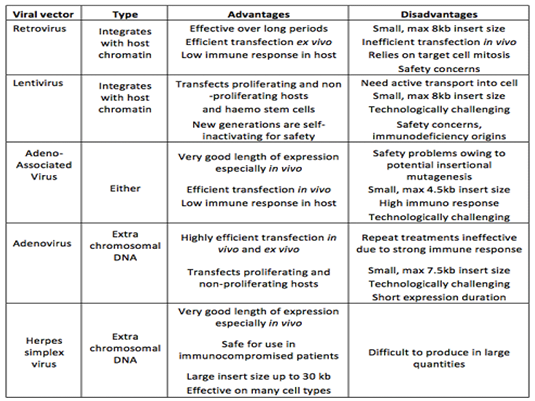
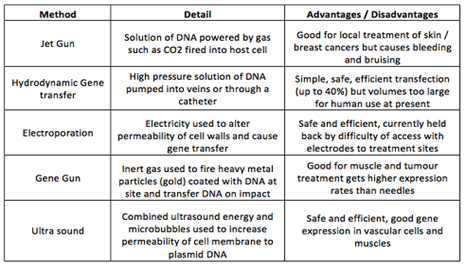
Vectors – To carry the transgene into the patient
Recombinant Virus à Recombining DNA materials by reassorting genetic segments
Plasmid DNA
- Therapeutic gene expression cassette embedded in a plasmid
- Liposomes:
- Containing DNA have been shown to undergo cellular
uptake through endocytosis
- Hybrid method:
- Virosomes à combine liposomes with an inactivated HIV or influenza virus.
- Shown to have more efficient gene transfer in respiratory epithelial cells than either
viral or liposomal methods alone
Target Diseases
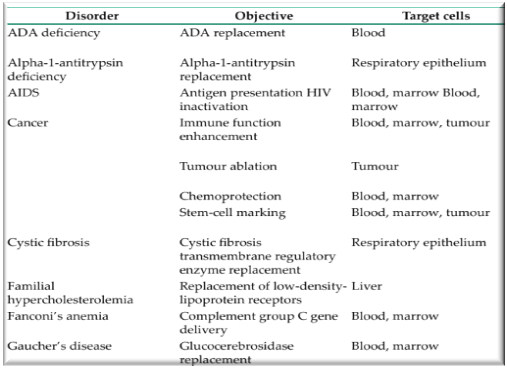
Leber’s Congenital Amaurosis
- RPE65 deficiency à photoreceptor-cell dysfunction
- A group of inherited, early-onset, severe retinal dystrophies
- Severe dysfunction of rod photoreceptor cells, which are wholly reliant on retinal pigment epithelium–derived RPE65, causes severely impaired night vision
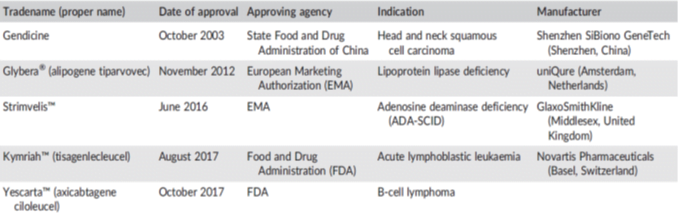
Approved Gene Therapy
- Gendicine (China in 2003)
- Oncorine (China in 2005)
- Neovasculgen (Russia in 2011)
- Glybera (Europe in 2012)
Gendicine
1st approved gene therapy in the world
- Recombinant Adenovirus
- E1 region replaced by wild-type p53 expression cassette – P53 à induce apoptosis, or stall the cell cycle
- Intra-tumoral injection (HNSCC)
- Tumour target cells infected
- Expressed p53 will exert anti-tumour effects
Oncorine
- First oncolytic genetically- modified virus approved (China – 2005)
- Recombinant Adenovirus Injection
- Deleting E1B55KD and E3 region of wild adenovirus
- Prevention of p53 degradation
- SCC of head, neck and oesophagus
- Combined with 5-FU and cisplatin
Neovasculgen
- Approved in Russia (2012)
- Peripheral arterial disease including critical limb ischemia (CLI)
- Gene of VEGF in a plasmid vector
- Stimulation of angiogenesis
Glybera
- Approved by EU (2012)
- Alipogene tiparvovec à AAV-1
- Familial lipoprotein lipase deficiency (LPLD) and suffering from severe or multiple pancreatitis attacks despite dietary fat restrictions
- Gain-of-function S447X variant of the human LPL gene (LPLS447X) in a AAV-vector
- Cost à $ 1.1 million per regimen!!!!

Tisagenlecleucel, marketed as Kymriah, is a treatment for B-cell acute lymphoblastic leukemia (ALL) which uses the body’s own T cells to fight cancer (adoptive cell transfer).
T cells from a person with cancer are removed, genetically engineered to make a specific chimeric cell surface receptor with components from both a T-cell receptor and an antibody specific to a protein on the cancer cell, and transferred back to the person.
The T cells are engineered to target a protein called CD19 that is common on B cells. A chimeric T cell receptor (“CAR-T”) is expressed on the surface of the T cell.
Disadvantages
- Short-lived nature [Cells à Long-lived and stable]
- Immune response [Foreign body response]
- Problem with viral vectors à [Toxicity/Immune response/ Targeting issues]
- Multi-genic disorders
- Insertional mutagenesis [Wrong DNA integration] – Leukemia due to rapid T-cell proliferation
Pricing Issues
- Market price of Glybera: US$ 1.1 million per treatment!!!
- Cost of preclinical development, manufacturing and distribution
- Personalised manufacturing and distribution
Ethical Considerations
- Deciding whether somatic gene therapy is more or less ethical than germ-line gene therapy
- Death of Jesse Gelsinger, an 18-year-old boy (Ornithine transcarbamylase deficiency)
- Died from multiple organ failure caused by a severe immune reaction in response to the use of first-generation adenoviral vectors
- Gene therapies use altered genes and potentially dangerous viruses, should treatments be tested more extensively?
- Will the high costs of gene therapy make it available only to the wealthy?
- Should people be allowed to use gene therapy to enhance basic human traits such as intelligence, or athletic ability?
Gene Doping
Gene therapy for restoring muscle lost to age or disease is poised to enter the clinic, but athletes are eyeing it to enhance performance. The non-therapeutic use of cells, genes, genetic elements, or of the modulation of gene expression, having the capacity to improve athletic performance is defined as Gene Doping by the World Anti-Doping Agency (WADA).
For example, gene doping could be used to provide athletes a source of erythropoietin (EPO), a hormone that promotes the formation of red blood cells that is already widely abused in sports.
Another candidate gene is Insulin-like Growth Factor 1 (IGF-1) which partly controls the building and repair of muscles by stimulating the proliferation of satellite cells.
