Reverse Pharmacology
Reverse Pharmacology (RP) is a science of integrating documented experiential hits, into leads by transdisciplinary exploratory studies and further developing into drug candidates by experimental research.
In this approach, the candidate travels a reverse path from ‘clinics to laboratory’ rather than classical ‘laboratory to clinics’.
- RP enhances the connection, communication and collaboration between modern sciences and technology with traditional medicine and modern biomedicine.
- It offers a major paradigm shift in drug discovery. Instead of serendipitous findings pursued randomly an organized path from clinical observations and success is established.
- Recently, India has amended the Drug Act to include a category of phytopharmaceuticals to be developed from medicinal plants by Reverse Pharmacology, with evidence of quality, safety and efficacy. These drugs will be distinct from traditional medicines like Ayurvedic, Unani or Siddha.
History of Reverse Pharmacology in India
Sir Ram Nath Chopra was a pioneer in the field of experimental pharmacology of indigenous drugs of India. Along with Gananath Sen he laid the foundation of RP of medicinal plants by pursuing clinically documented effects of Ayurvedic drugs.
Sen and Kartik Bose in 1931 demonstrated the antihypertensive and tranquilizing effect of Rawolfia serpentina.
Scope of Reverse Pharmacology
- to understand the mechanisms of action at multiple levels of biology and to optimize safety, efficacy and acceptability of the leads in natural products based on relevant science.
- Scientifically validated and technologically standardized botanical products may be explored on a fast track using innovative approaches like reverse pharmacology and systems biology, which are based on traditional medicine knowledge.
Salient features of reverse pharmacology:
- Classical drug discovery process is an expensive and time-consuming process. It involves 10 – 15 years of investigation period and investments of the order of US $ 1 to 1.5 billion. This extremely complex, technology bases and capital-intensive process has resulted in ‘target rich lead poor’ performance. Whereas Reverse Pharmacology is an economical, time sparring and has least bottlenecks. It effectively reduces the duration of drug development to 4 – 5 years.
- It also allows understanding the mechanisms of drug action at multiple levels and helps in optimizing the safety, efficacy and acceptability of the leads from the natural products.
- RP utilizes traditional knowledge of medicines to discover drugs and is also called as a path of pharmacology from the bedside to bench experiments.
- In this process ‘safety’ remains the most important starting point and the efficacy becomes a matter of validation.
The path of classical and reverse pharmacology approach:
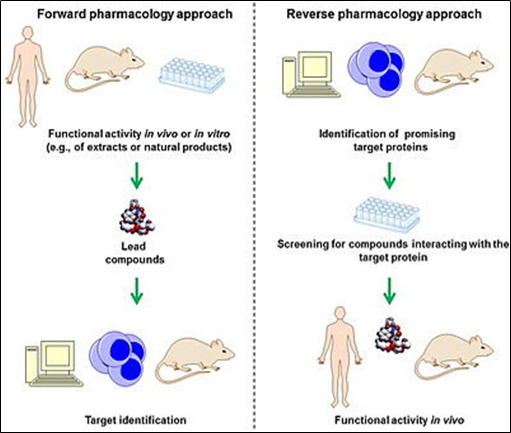
Research and development path for natural products:
The different paths of development of botanical medications are:
- The ethno medicine path – Based on observing field use of plants and then standardizing for the activity e.g. tubocurarine isolated from Curare, commonly known as South American arrow poison.
- Through screening of extracts in selected models, isolation of the active principles and then definition of a single molecule, for preclinical and clinical evaluation such as reserpine from Rauwolfia serpentina.
- The herbal product path of screening of extracts in models and then testing for efficacy and safety in a clinical set up such as Ganoderma lucidum and Hypericum perforatum.
- The holistic path of using the formulation mentioned in classical texts of Ayurveda and then evaluating efficacy of popular medicines in use such as chavanprash.
The drawbacks of these botanical medications are:
- Although botanical medications continued to be produced in every country, their clinical efficacy was usually not evaluated and the composition of these complex mixtures was only crudely analysed. Investments in these methodologies remained scarce.
- Failure to distinguish folklore from traditionally established systems like Ayurveda and TCM (Traditional Chinese Medicine).
- Lack of proper identity and implementation of Good Laboratory Practices, improper experiential documentation.
- Absence of Phase II, dose optimizing studies.
- Cultural prejudice for modern medical sciences.
- Lack of political and financial support. Plants are compositional complex mixtures of chemicals and studying them is a challenge.
Reverse pharmacology provides an open – ended approach, to guide us from the traditional wisdom of Ayurveda to the current state-of-art developments in biology. It offers a major paradigm shift in drug discovery. Instead of serendipitous findings pursued randomly, an organized path from clinical observations and successes is established.
R & D path for natural products:
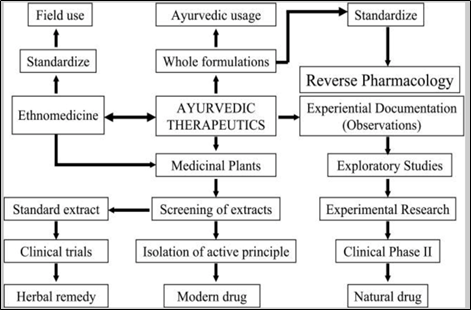
Reverse Pharmacology and Ayurveda:
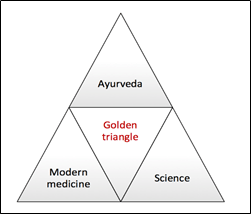
Ayurvedic knowledge and experiential database can provide new functional leads to reduce time, money and toxicity – the three main hurdles in the drug development.
Basic principle of Ayurveda is based on personalized approach can be used for creating personalized, customized or designer medicines. Therefore, science of Ayurveda has the potential to revolutionize modern medicine and drug discovery processes.
A golden triangle consisting of Ayurveda, modern medicine and science will converge to form a real discovery engine that can result in newer, safer, cheaper and effective therapies.
The salient features of drug discovery by reverse pharmacology through Ayurveda are:
- The search is based on Ayurvedic medicine research, clinical experiences, observations or available data on actual use in patients as a starting point.
- Use of principles of systems biology where holistic yet rational analysis is done to address multiple therapeutic requirements.
- Since safety of the materials is already established from traditional use track record, we undertake pharmaceutical development, safety validation and pharmacodynamic studies in parallel to controlled clinical studies.
The approach has attracted considerable attention, nationally and internationally: Central Scientific Instrumental Research (CSIR) and Indian Council of Medical Research (ICMR) have done clinical trials with natural products. Moreover, Central Council for Research in Ayurveda and Siddha (CCRAS) has recently adopted the golden triangle approach for some new indications of old drugs, as well as for Ayurveda.
A New Millennium for Technological Leadership Initiative (NMITLI) has resulted in hits, leads and effective formulations for diabetes, arthritis and hepatitis with novel mechanism of action and intellectual property as shown in the following table.
Phases of reverse pharmacology:
RP can be perceived to comprise of three phases or domains.
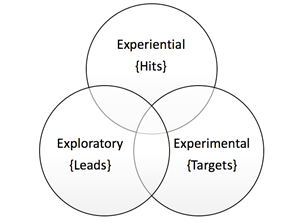
- Experiential phase that includes robust documentation of clinical observations of the biodynamic effects of standardized Ayurvedic drugs by meticulous record keeping.
- Exploratory studies for tolerability, drug-interactions, dose-range finding in ambulant patients of defined subsets of the disease and para- clinical studies in relevant in vitro and in vivo models to evaluate the target-activity.
- Experimentalstudies, basic and clinical, at several levels of biological organization, to identify and validate the reverse pharmacological correlates of Ayurvedic drug safety and efficacy.
The progress of RP in India has led to the following leads from Ayurvedic medicinal plants:
| Disease | Plants | Ayurvedic forms | Phytoactives |
| Diabetes mellitus | P. emblica E. littorale | Fruits-Chyavanprash Entire plant ghana | Gallic acid Swertiamarin |
| Arthritis | T. cordifolia S. anacardum | Amrita bhallatak Avaleha | Polysaccharide Bhilawanol |
| hepatitis | Multi-ingredients | Phalaghrita | Multiple |
| Psoriasis | A. mexicana | Latex ghana | Berberine |
| NASH | P. karroa | Powder | Picroside 2 |
| Malaria | N. arbor-tristis | Leaves-paste | unknown |
| Pre-cancer Lesions | C. longa | Powder | Curcumin |
Thus, the Reverse pharmacology as an organized trans-discipline would fill up the gaps between the serendipity and research creativity.
