Evaluation of Diabetes

Insulin – Discovered by Banting& Best
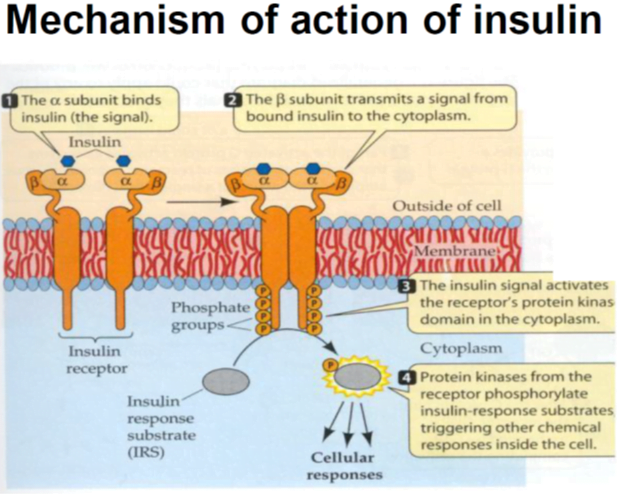

Need for new drugs
- Only one-third of patients diagnosed as having diabetes achieve the American Diabetes Association goal of an HbA1c level of less than 7%
- A high degree of compliance mandatory on the part of the patients
- Therapy does not prevent or reverse complications of diabetes
- Exogenous insulin
- fails to mimic physiologic insulin peaks and troughsInjections are inconvenient
- Oral hypoglycemic agents
- Unpredictable hypoglycemia
- Drug- specific toxicity
In-vitro:
Effect on pancreas:
The isolated pancreas of rats:
Purpose: the effect of the drug on insulin, glucagon & somatostatin secretion without interference from other organ systems.
Procedure: Pancreas of Wistar rats isolated and perfused with PSS and glucose. Test compound in different concentrations added to perfusate, f/b glucose. The perfusate is collected every minute for 30 min to measure the level of the hormones.
Variable: Values of each hormone, for each minute, are plotted versus time. The effect of test compounds on hormones in response to elevated glucose is compared with the control.
Isolated pancreatic islets:
Purpose: To study their dynamic response between various metabolic states
Procedure: Islets isolated from Wistar rats by collagenase method, perfused with PSS & glucose. For 2-18 min, increasing concentrations of test compound added f/b simultaneous increase in glucose concentrations.
Variable: measure the level of insulin.
Receptor binding studies:
Procedure: Insulin-producing tumor cells are used (RIN-m, INS-1 cells) à
These are incubated with [3H] glibenclamide or [3H] glimepiride (or any appropriate makers e.g for Incretin receptors) which bind to specific receptors. à Then test drugs are added
Variable: displacement of the labeled marker by test drug is noted by liquid scintillation counters.
Effect on the liver:
Perfusion of isolated rat liver:
Purpose: to study the effect of the drug on net glucose production and lactate utilization by the liver.
Procedure: Liver of Wistar rats isolated and perfused with PSS & Na- L- Lactate for 5min. The test compounds are added in increasing concentrations to the perfusate medium. The system is run for 2 hrs and samples are collected at intervals.
Variable: Net glucose production from lactate & net lactate utilization is determined from the effluate.
The values are plotted against time before and after the addition of test compounds
In-vivo:
Experimental models are developed with the purpose of enhancing understanding of the pathophysiological mechanisms of diseases that affect humans.
Regardless of the classification, the resulting metabolic abnormalities that characterize diabetes contribute greatly to the clinical complications, and the major clinical strategy is aimed at restoring metabolic balance. However, a major concern in testing potential and successful interventions in humans is that due to the natural history of T2D, it takes years for the complications to develop.
Advantages
- Interventions can be assessed in a shorter time- a span in representative animal models.
- Allows investigation of pathophysiology and evaluation of potential strategies for treatment & prevention of the disease and related complications.
- To circumvent the ethical and logistical constraints inherent in studying diabetes in the human population exposed at random to chemical and microbiological agents, investigators continue to rely on animal models that can be readily tested, biopsied, and autopsied.
- It is possible to breed these models to study and manipulate inheritance and to test for their response to environmental agents.
- Most of the available models are based on rodents, because they are small, easy to handle, and economically advantageous, and have a short generation interval.
Disadvantages-
To be relevant, an animal model should mirror the pathophysiology and natural history of diabetes or the model should develop complications of diabetes with an etiology similar to that of the human condition. They only mirror specific conditions as seen in humans.
Type 1 DM:
- Chemically induced type 1 diabetes
- Spontaneous autoimmune models of type 1 diabetes
- Virus-induced models of diabetes
- Non- rodent models of type 1 diabetes
Chemically induced type 1 DM:
PRINCIPLE: I
In chemically induced models of type 1 diabetes, a high percentage of the endogenous beta cells are destroyed, and thus, there is little endogenous insulin production, leading to hyperglycemia and weight loss.
- Diabetes is usually induced around 5–7 days prior to the start of the experiment to ensure stable hyperglycemia.
- Streptozotocin (STZ) or Alloxan
- Chemically induced diabetes is appropriate to use when testing drugs or therapies where the main mechanism of action in lowering blood glucose in a non-beta-cell-dependent manner; for example, to test new formulations of insulin
- Single high dose:
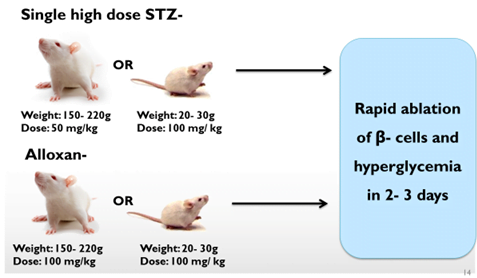
Alloxan produces a triphasic response. New formulations of insulin can be tested in this model.
Blood glucose levels show triphasic response with hyperglycemia for 1 hour f/b hypoglycemia that lasts for 6hours & stable hyperglycemia after 48hours.
- Multiple low doses:

Treatments that may prevent beta-cell death can be tested in this model.
- Advantages: Simple, cheap and selective loss of beta cells without affecting other cells of the pancreas.
- Disadvantages: Toxic to other organs. Animals might die, Cells might regenerate. Variable hyperglycemia might develop. Guinea pigs and rabbits are resistant to the action of STZ
| Possible uses of the model: | |
| Single-dose models | New formulations of insulin |
| Multiple STZ dose model | Treatments that may prevent beta cell death |
- Spontaneous autoimmune DM1 models:
These are useful in understanding the Pathophysiology of DM1 and studying immunomodulatory interventions.
| NOD mouse (Non- obese diabetic) | BB rats | LEW.1AR1/iddm rats | |
| Age at onset of diabetes | Insulitis at 3- 4 wks f/b overt diabetes at 10-14 wks up to 6 mo) | Insulitis and insulin deficiency at 8- 16 wks | Insulitis for at least a week prior to onset at 8- 9 wks |
| Insulin deficiency | Mild | Severe, require insulin for survival | Severe |
| Gender effect | M < F | M = F | M = F |
| Associated autoimmune diseases | Sialadenitis, thyroiditis | Thyroiditis | Organs free of infiltration |
- Virus induced DM1:
Encephalomyocarditis virus, Coxsackie virus.
| Virus | Peculiarities of disease |
| D- a variant of encephalomyocarditis virus (EMC- D) | Selectively infects and destroys pancreatic β- cells in adult male ICR- Swiss mice Adult male C3H/ HeJ mice are resistant Pretreatment with cyclosporine- A : Increased severity and incidence of diabetes in ICR- Swiss mice and induction of diabetes in C3H/ HeJ mice |
| Diabetogenic strain (E2) of coxsackievirus B4 [Coxsackie virus-> CD I mice (C for C)] | CD 1 mice- Islet cell destruction was associated with chronic islet cell inflammation, the elevation of islet cell antibody, the prolonged presence of viral DNA in the pancreas |
- Non-rodent models:
PANCREATECTOMY:
- Dogs
- Pigs
- Primates
- Skilled
- Disadvantages
- Invasive method
- Chances of hypoglycemia
- Leads to the pancreatic exocrine deficiency in the animal
- However, pancreatectomy in pigs followed by auto-transplantation of the isolated islets is a reasonably accurate model of auto-transplantation of islets in humans
- Hormone induced –
- Growth hormone-induced diabetes
- Dogs and cats after repeated administration of GH exhibit the severe form of diabetes
- Rats are resistant
- Corticosteroid induced diabetes
(Dexamethasone)
- Dexamethasone (2-5mg/kg i.p. BD over a number of days in rats)
- Growth hormone-induced diabetes
Type 2 DM:
- Obese models of type 2 diabetes
- High-fat feeding
- Non- obese models of type 2 diabetes
- Non- rodent models of type 2 diabetes
Type 2 diabetes is characterized by insulin resistance and the inability of the beta cell to sufficiently compensate.
Therefore, animal models of type 2 diabetes tend to include models of insulin resistance and/or models of beta-cell failure. Many animal models of type 2 diabetes are obese, reflecting the human condition where obesity is closely linked to type 2 diabetes development.
- Monogenic obese models:
- Although obesity in humans is rarely caused by a monogenic mutation, monogenic models of obesity are commonly used in type 2 diabetes research.
- The most widely used monogenic models of obesity are defective in leptin signaling.
- Leptin induces satiety, and thus, a lack of functional leptin in these animals causes hyperphagia and subsequent obesity.
- Lepob/ob mice: Spontaneous mutation in C57BL/6 mice.
- Leprdb/db mice:
- Zucker diabetic fatty rats: Mutation in leptin receptor of ZFR. Diabetic complications can be studied.
- All the models develop hyperglycemia and hyperinsulinemia.
These models can be used for studying treatments to improve insulin resistance and treatments to improve β- cell function.
- Polygenic models:
These are more accurate models of human DM2.
- Useful in
- studies that have aimed to reverse the symptoms of type 2 diabetes
- understand more about the interplay of obesity and glucose homeostasis
- study diabetic complications
- KK- Ay mice: derived by introducing the yellow obese gene into the KK strain, Severe hyperinsulinemia and insulin resistance in both muscle and adipose tissue
- NZO mice: Hyperphagic, obese, hyperleptinemic by 9- 12 wks of age.
- OLETF rat: Spontaneously diabetic derived from Long- Evans Rats. It exhibits renal complications.
- These models can be used for studying treatments to improve insulin resistance and treatments to improve β- cell function.
High-fat feeding:
- Normal rat chow is Replaced by a diet where 58% of energy is derived from fat
- C57BL/6 mice fed with a high- fat diet for several weeks to induce pronounced weight gain
- Weight gain associated with insulin resistance and impaired glucose tolerance due to lack of compensation by β- cells
- Obesity-induced by environmental manipulation rather than genes- more accurately models human DM
Non- obese models of type 2 diabetes
- Not all type 2 diabetes patients are obese, and thus, it is important that lean animal models of type 2 diabetes are also studied.
- Goto- Kakizaki rats (GK)
- Lean model of type 2 DM
- Characterized by glucose intolerance and defective glucose-induced insulin secretion
- Defective glucose metabolism due to aberrant β- cell mass and/ or function- as volume & density of β- cells similar to controls
Possible use
Treatments to improve β- cell function
Treatments to improve β- cell survival
In-vivo assay for insulin and insulin-like activity:
Blood sugar lowering in rabbits: Rabbits are fasted for 24 hrs and then injected s.c with test/control. Glucose levels in venous blood estimated at 1 hr and 2.5 hrs post-injection.
Hypoglycemic seizures in mice: Biological standardization of insulin using the mouse convulsion method. Mice fasted for 20 hrs. Test/control administered and the number of mice is recorded that is dead, convulse or lie still for more than 2 or 3s when placed on their backs.
Models of beta-cell manipulation: highly relevant in understanding pathways that can lead to the inability of beta cells to secrete appropriate amounts of insulin.
- Mutations in Kir6.2 (which cause permanent neonatal diabetes) to study KATP channel function- studied in mice
- Mutations in genes encoding glucose kinase to understand the function of a glucose sensor in β- cells (childhood-onset NIDDM)
Beta-cell regeneration models: β cell mass tends to be partially or nearly completely ablated by a variety of means, & mechanisms of beta-cell mass renewal are investigated.
Neonatal STZ administration: STZ (100 mg/kg i.v. or i.p.) given to 2 day old rats- regeneration/ type 2 diabetes model in adults- f/b regeneration of β- cell & normoglycemia by day 10. Hyperglycemia returns by 6 wks due to inadequate β- cell mass & dysfunction
Regeneration using genetic approaches: β- cells are genetically modified to express the DT(Diphtheria toxin) receptor under the rat insulin promoter. On administration of DT, β- cells are selectively ablated. 4- 17% of the β- cell mass regenerates in 10 months
Study of diabetes in pregnancy
- Pups born from rats rendered diabetic by STZ treatment are more
likely to become diabetic in adult life than pups from rats not receiving STZ.
Other parameters evaluated in this model include-
- Mean number of pregnancies reaching term
- No. of pups LGA and SGA etc
Endpoints to study in animal models of DM:
- Non- fasting blood glucose concentration over 250 mg/dL or preferably a chronic elevation over 300 mg/dL is appropriate to consider a mouse diabetic.
- Detection of glucose in urine.
- Weight gain/loss.
- GTT.
- Serum insulin or C-peptide levels.
- Pancreatic histology.
Clinical evaluation:
Phase 1:
Objective: Safety, tolerability, PK and PD.
Subjects: Normal healthy volunteers or patients with DM. It might be necessary to study patients with DM since there are significant changes in drug disposition and drug absorption parameters.
Endpoints: PK (Cmax, AUC, T max t1/2), glucose metabolism (serum glucose, insulin levels C peptide levels), Safety (BP, BW, Hemat parameters)
Phase 2 DM1:
Design: Wide Intra and inter-subject variability: Crossover designs may be preferable. However, with drugs having a long duration of action cross overstudies are not preferable d/t carry-over effect, then do parallel studies.
Duration: 4 weeks for cross over, 3 months for parallel.
Endpoints: same
Phase 3 DM1:
Design: Parallel Studies are active-controlled using an insulin preparation as a comparator with a pharmacological profile similar to that of the tested agent
Duration: 6 months and follow-up of 12 weeks.
Endpoints: same
DM2: All these studies should have a run-in period before randomization (6-8 weeks) for diabetes education, optimization of compliance of diet and exercise, stabilization of HbA1C and to screen out non-compliant subjects.
Placebo-controlled monotherapy trials:
Subjects: Drug naïve subjects whose diabetes is well controlled with diet and exercise or subjects on low dose therapy who can be withdrawn from t/t.
Endpoint: a difference between active drug and placebo (or between two active treatments such as a lower and higher dose of the test agent) in the proportion of subjects meeting criteria for glycemic rescue therapy.
- Placebo-controlled add-on therapy trials:
- Subjects: Patients inadequately controlled on optimal or near-optimal doses of approved therapies- IND or placebo added to the existing therapy
- Endpoints: Reduction in HbA1C. Change in plasma glucose, GTT, the effect on BP. Lipids, BW, BMI, etc. Safety: incidence of hypoglycemia, local reaction to injections and product immunogenicity.
Microvascular endpoints: The clinical benefit of improved glycemic control may not be evident until several years later. Thus, extended follow-up is required to adequately assess the microvascular or macrovascular effects of an intervention.
MACE (Major adverse cardiovascular events) and mortality endpoints: FDA guidance Diabetes Mellitus—Evaluation of Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes. Non-fatal myocardial infarction, non-fatal stroke, and cardiovascular mortality. Large and longer phase 2 and 3 trials are recommended to assess these outcomes. Upon completion of a planned preapproval clinical development program, a meta-analysis exploring the investigational drug’s MACE liability is to be conducted.
- CI > 1.8: It is unacceptable. an additional single, large safety trial should be conducted that alone, or added to other trials, would be able to satisfy this upper limit of the CI.
- CI between 1.3 and 1.8: A post-marketing trial is required to definitively show that the upper limit of the CI is actually less than 1.3
- CI is <1.3: a post-marketing cardiovascular trial generally may not be necessary.
